 Sodium is largely used in making NaCl, Na 2CO 3, NaOH and other useful compounds.Here are some uses of the sodium element. Sodium forms various compounds like NaCl, Na 2CO 3, NaOH, NaNO 3, etc.The sodium has an electron configuration 3s 1 and it has only 1 valence electron.When sodium metal is burnt in a flame, it produces a golden-yellow flame.But it is always found in the compound with other elements. As sodium is a reactive metal, it is not found in free state. 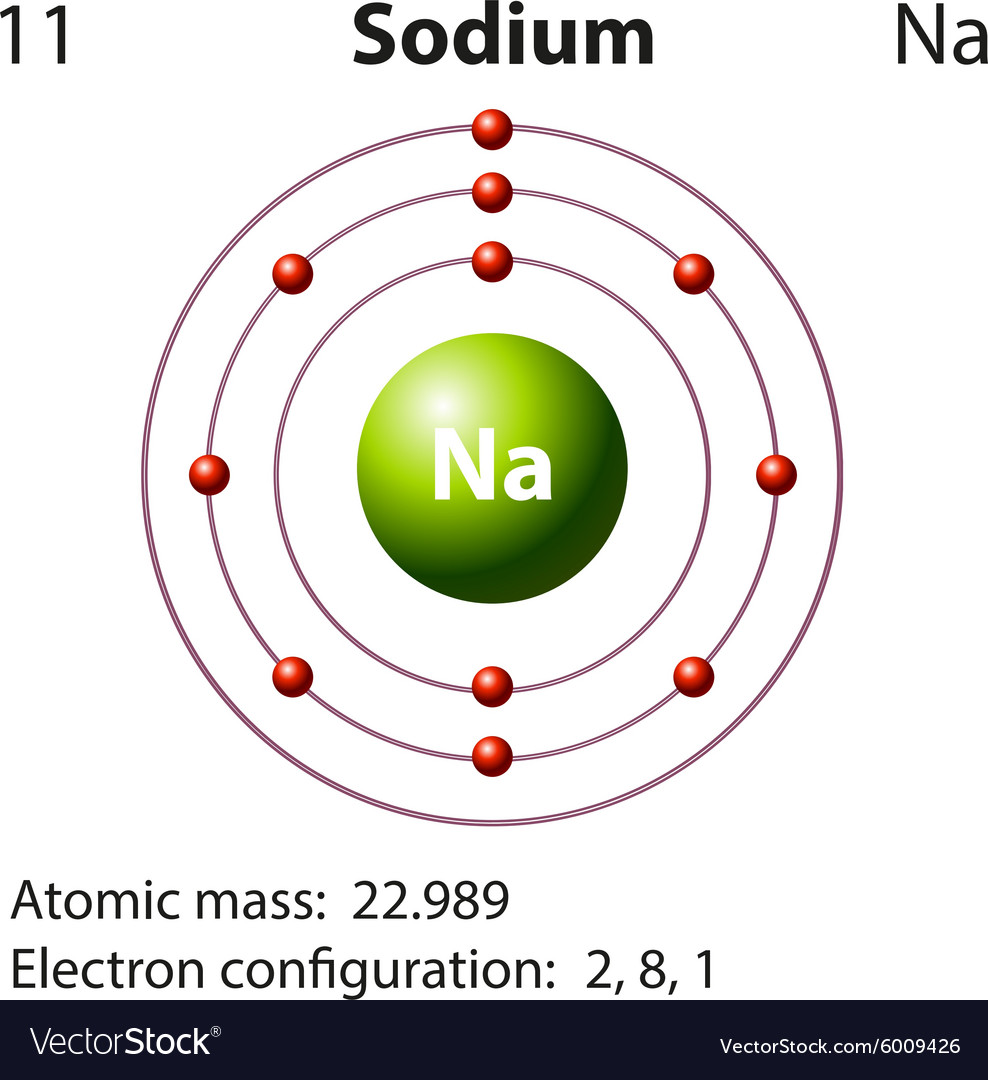
Sodium is a reactive metal and it forms an oxide layer if kept open in the air.There are many isotopes of sodium, but out of those isotopes, the most stable isotope is 23Na.The melting point and boiling point of sodium are 97.7 ☌ and 882.9 ☌ respectively.Sodium metal is a good conductor of electricity as it has only 1 valence electron.It is soft metal and it can be cut with a knife.Sodium is a soft metal having a silvery white appearance.Here is a list of some physical properties and chemical properties of sodium. Around 2.3% of the earth’s crust contains sodium element.Sodium is the 6 th most abundant element found from the earth’s crust.Sodium is the most abundant alkali metal.Sodium is also present in the human body, and it maintains the fluid balance in the body cells.Sodium floats on water because it has less density than that of water.Sodium is a soft metal and it can be cut with a knife.Here are a few interesting facts about sodium element. H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La* Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac** Rf Db Sg Bh Hs Mt Ds Rg Cn Nh Fl Mc Lv Ts Og * Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu ** Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No LrĬlick on above elements in the periodic table to see their information. Appearance of sodium Silvery white Atomic number of sodium 11 Symbol of sodium Na Atomic mass of sodium 22.990 u Protons, Neutrons & Electrons in sodium Protons: 11, Neutrons: 12, Electrons: 11 State of sodium (at STP) Solid Group number of sodium in periodic table 1 Period number of sodium in periodic table 3 Block of sodium in periodic table s-block Category of sodium Alkali metal Bohr model or Electrons per shell or Electrons arrangement in sodium 2, 8, 1 Electron configuration of sodium 3s 1 Orbital diagram of sodium Valence electrons in sodium 1 Electronegativity of sodium (on pauling scale) 0.93 Atomic radius of sodium (van der Waals radius) 227 picometers Density of sodium 0.97 g/cm 3 1st ionization energy of sodium 5.139 eV Main isotope of sodium 23Na Melting point of sodium 370.9 K or 97.7 ☌ or 208 ☏ Boiling point of sodium 1156 K or 882.9 ☌ or 1621.2 ☏ Crystal structure of sodium Body Centered Cubic (BCC) Discovery of sodium By Humphry Davy in 1807Īlso see: Interactive Periodic Table (It has rotating bohr models as well as many other details of all the 118 elements in a single periodic table). The important data related to sodium element is given in the table below. Uses of Sodium Sodium Element (Information Table).  
There is a lot more information related to sodium which is mentioned in the Information Table given below. Sodium is in the s-block and it is classified as an alkali metal on the periodic table. Sodium element (Na) is in group 1 and period 3 of a periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
